Gas Laws Chapter 5. Pressure Force per unit area Measured in Atmospheres ( atm) Mm of Hg = Torr Pascals or kiloPascals (Pa or kPa) - ppt download

Which of the following is/are correctly matched ? Ar atm > 1 torr > 1 Pa → (Units of pressure) (B) SO2- > N2 > 02- → (Number of electrons per species) (

pressure, conversion units into defferent units,atm,bar,torr,psi,Pascal,mmHg, numerical,and examples - YouTube

Pressure Conversions: 1 atm = 760 mmHg = 760 torr = 101.3 kPa = 14.7 psi Use the conversion factors above - brainly.com

Pressure 1 atmosphere ~ 1 bar ~ 760 mm Hg ~ 760 torr ~ 100,000 Pa Ion gauges read in mbar i.e. 1x mbar = 1x atm. Sometimes ion gauges read. - ppt download

Pressure Conversion Practice Problems (atm to torr, atm to mm Hg, atm to kPa, torr to kPa, etc) - YouTube

What is the value of 1 atm in torr. Defineatmospheric pressure. | 9 | STUDY OF GAS LAWS | CHEMI... - YouTube

Converting Pressures 760 mm Hg = 760 torr 1 atm = 760 mm Hg 1 atm = kPa 760 mm Hg = kPa Converting Pressures: Convert 740 torr to kPa ppt download

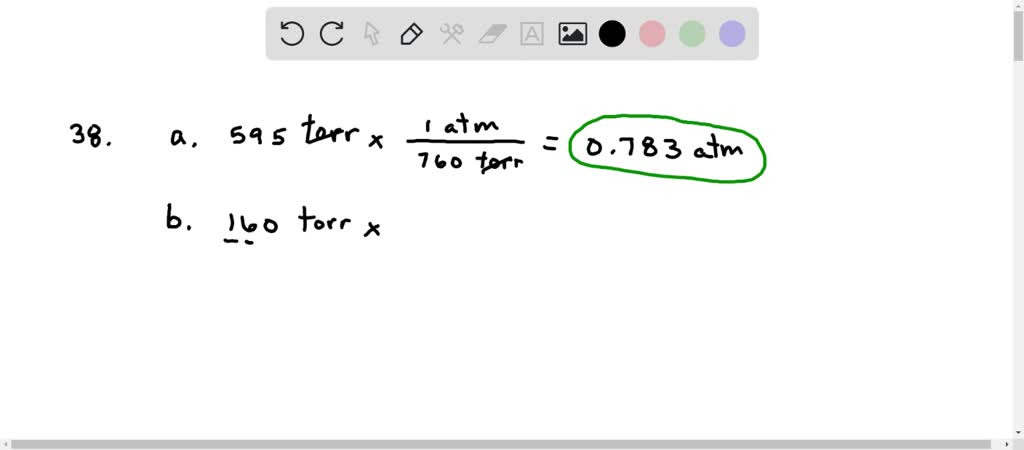
SOLVED:Carry out the following unit conversions: (a) 0.625 atm to torr, (b) 825 torr to atm,(c) 62 mm Hg to torr (d) 1.22 kPa to bar.

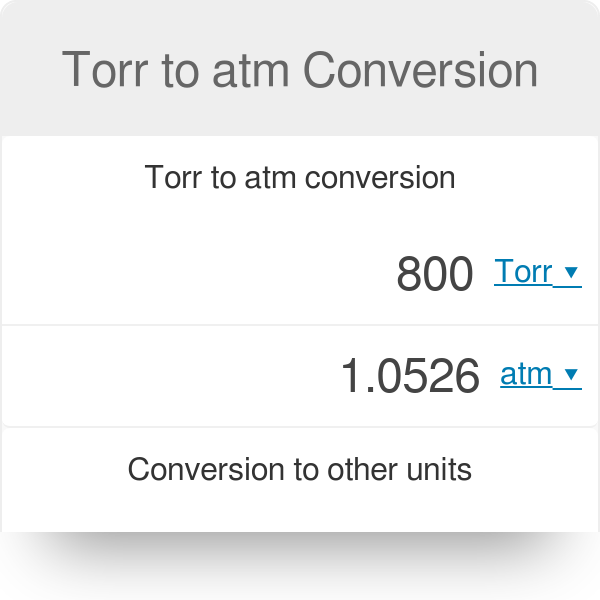
SOLVED: Conversion Factors: 1 gal = 231 in³ (exact) 1 atm = 760 torr (exact) 1 cm³ = 1 mL (exact) 1 lb = 453.59237 g (exact) 2.54 cm = 1 in (